Our laboratory examines the structure and function of synapses in the mammalian neocortex and their contribution to complex circuit activity and behavior. We are particularly interested in applying an array of methodological approaches, including electrophysiology, 2-photon imaging and transmitter photo-uncaging, optogenetics, and viral tracing to both reduced preparations and intact behaving animals. In this way, we hope to bridge the gaps between molecular, cellular, and systems neuroscience.
Development, function, and plasticity of inhibitory GABAergic circuits.
The balance of synaptic excitation and inhibition is thought to be critical for normal brain function and is disrupted in a variety of neuropsychiatric disorders. In the neocortex, this balance is maintained by an intricate dance between excitatory glutamatergic pyramidal neurons and inhibitory GABAergic interneurons. A major challenge to understanding the role of GABAergic inhibition is the incredible diversity of interneurons, with different subtypes defined by molecular, electrophysiological, and anatomical features corresponding to distinct functions in local microcircuits. In our studies, we use acute brain slice preparations to dissect the organization of GABAergic synapses and their ability to regulate postsynaptic activity. We have focused particularly on inhibition targeting pyramidal neuron dendrites (see below), which influences both electrical and biochemical signaling in the postsynaptic cell. We are also using in vivo approaches to explore the role of different interneuron populations and inhibitory synapses in the control of learning and perception.
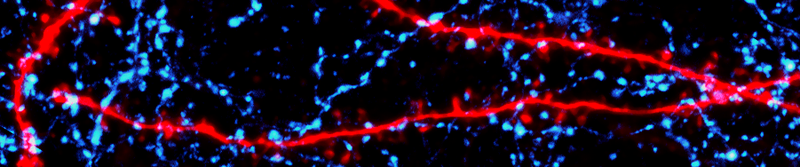
Pyramidal cell dendrites (red) interwoven with somatostatin-expressing interneuron axons (blue) in the mouse neocortex.
Cortical microcircuits underlying visually-guided behavior.
Visual information is encoded by neuronal activity in the visual cortex, whose diverse anatomical projections route these signals to various downstream locations that subserve different aspects of perception, learning, and motor output. Outputs from pyramidal neurons in Layer 5 form the major pathway by which cortical information is communicated to subcortical structures, including the basal ganglia, superior colliculus, and brain stem. We are using novel viral tracing approaches to understand the organization of these projections, combined with 2-photon and mesoscopic imaging in vivo to track the activity of identified neurons during the performance of visually-guided behaviors. We are also developing novel imaging methods to enable simultaneous monitoring of local and large-scale circuits in the awake, behaving animal (see below).

Simultaneous whole-cortex mesoscopic (left) and local 2-photon (right) imaging of GCaMP6 in the awake mouse.
Neuromodulation: providing functional flexibility to cortical circuits.
Adaptive behavior over the life of an organism requires a nervous system with sufficiently stable wiring to support long-term memory but plastic enough to adjust to rapid changes in environmental context. Much of this dynamic flexibility is provided by neuromodulators such as norepinephrine and acetylcholine, which influence neuronal excitability and synaptic transmission. We are using a combination of approaches in both brain slices and behaving mice to study the actions of neuromodulation on identified microcircuits in the mouse visual cortex.
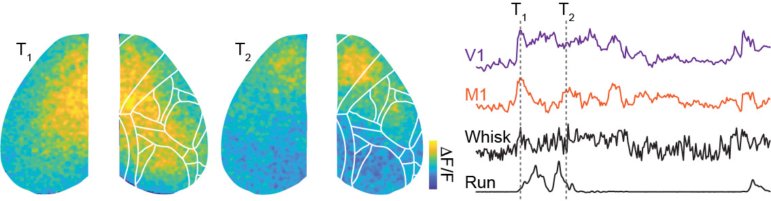
Mesoscopic acetylcholine imaging in the awake mouse, using fluorescent GRAB reporters (courtesy of Yulong Li Lab). Signals are heterogeneous across cortical regions and correlated with behavior.
Models of neuropsychiatric illness.
A large body of evidence now suggests that disruption of synaptic transmission and subsequent dysfunction of neuronal circuits contributes to the pathophysiology of neuropsychiatric disorders such as schizophrenia and autism. We are actively investigating how genetic mutations of disease-linked genes, including MeCP2 (Rett Syndrome) and Rai1 (Smith-Magenis Syndrome) alter the function and plasticity of cortical synapses and produce consequences for behavior. In recent work, we are taking advantage of novel viral strategies to induce whole-brain CRISPR/Cas9-mediated deletion of targeted genes in postnatal animals.
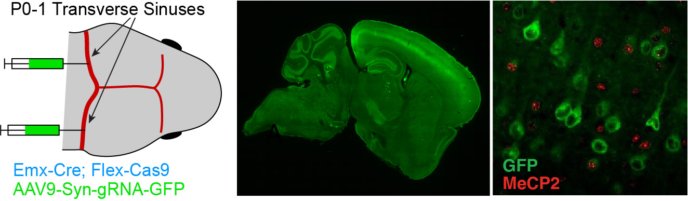
Whole-brain expression of guide RNA following neonatal injection of AAV9 in a Cas9 transgenic mouse drives cortical deletion of MeCP2.